
News
News updates from the Trellis Bioscience Team.
August 2024
August 4, 2024
Significance of Biofilm Disruption — Trellis Bioscience's TRL1068: Advancing a New Class of Anti-Infective Drugs
(View Article)
In the natural world, biofilm is a complex aggregate of bacteria and a bacterially produced extracellular matrix that provides a protective environment for the microorganisms, shielding them from natural antibiotics and acute environmental stressors such as nutrient restriction, desiccation, UV radiation, extreme temperature, and pressure, and high alkalinity or acidity. Biofilms affect industrial pipes and underwater structures and are ubiquitous in soil [1].
In the human body, bacteria in biofilms are protected from attack by pharmaceutical antibiotics and by the immune system, allowing persistence even in hostile conditions. Within the biofilm matrix, bacterial cells adopt a defensive physiological state that includes coordinated expression of multiple resistance genes [2].
Trellis Bioscience's TRL1068 is the first clinical-stage agent for a new class of drugs called biofilm disruptors [6]. Clinical proof-of-concept for TRL1068 was achieved in patients with periprosthetic joint infections (PJI) [7]. Lower extremity (knee and hip) total joint arthroplasties are among the most commonly performed surgical procedures in the USA, providing a substantially improved quality of life for most patients. However, despite advances in perioperative antiseptic protocols, surgical technique, and operating room sterility, infection remains the most common cause of prosthetic joint failure within the first year after primary arthroplasty [8], affecting ~2% of patients.
July 2024
July 31, 2024
Industry Disruptors: These Companies Are Leading the Way
(View Article)With the influx of technological advancements and evolving customer expectations, some companies are not just a part of their industry; they are completely disrupting it. These companies are making a difference with their innovative approach and bold business model. From transforming healthcare to helping build a more sustainable future, these companies are leading the way.
Trellis Bioscience is a therapeutic antibody company developing treatments for infectious diseases that are currently undergoing clinical studies. Bacterial biofilm is always present in chronic Prosthetic Joint Infections, which makes the use of antibiotics ineffective. As a result, patients must replace the infected prostheses with new ones through complex and costly surgeries. With its proprietary CellSpot™ platform to identify monoclonal antibodies from the blood of healthy human donors, Trellis identified TRL 1068 as a potential therapy. When infused, TRL1068 disrupts the bacterial biofilm barrier, allowing antibiotics to kill bacteria. Hopefully, in the future, there will be no need for major surgeries in PJI patients, just TRL1068 and antibiotics. “Our body can deal with small amounts of bacteria. But when there’s a massive biofilm growing, that is a problem. We make antibiotics work again. That is a huge game-changer not only in PJI patients,” explains CEO Stefan Ryser.
July 16, 2024
Trellis Bioscience's TRL1068 Study Published in AAC: A Breakthrough in AMR
Trellis Bioscience, Inc. is excited to announce the peer-reviewed publication of the results of a landmark clinical
study in a leading infectious disease journal, Antimicrobial Agents and Chemotherapy (AAC), from the American Society of Microbiology on July 16, 2024. The study
highlights significant advancements in treating biofilm-associated infections using TRL1068, a human monoclonal antibody that disrupts bacterial biofilm. The results highlight the potential impact of this innovative agent in combating
antimicrobial resistance (AMR).
The article, entitled "Phase 1 study of the pharmacokinetics and clinical proof-of-concept activity of a biofilm-disrupting human monoclonal antibody in
patients with chronic prosthetic joint infection of the knee or hip” shows that TRL1068 is well tolerated, with favorable biodistribution. In addition,
the study showed substantial reductions in biofilm in treated patients, including the complete elimination from the infected prostheses of three patients.
Biofilms, complex and naturally occurring communities of bacteria protected by an extracellular matrix, are a major obstacle in treating drug-resistant bacterial
infections. Conventional antibiotics typically fail against biofilm-embedded bacteria due to reduced access and altered bacterial physiology.
(View Press Release)
July 11, 2024
Native human monoclonal antibody (mAb) for H5N1
To date, H5N1 influenza has been confirmed in 850 people, with 60% mortality in those people. It is now expanding in dairy cattle across the US, with only a few mutations needed for it to become a pandemic worse than COVID-19. Since even the best influenza vaccines fail to induce protective immunity in a substantial fraction of subjects, and are a slow response to an evolving threat, an acute treatment is needed as a complementary intervention to forestall the worst outcomes. Trellis’s proprietary technology for cloning mAbs from human white blood cells has been validated by a clinical trial of a mAb to overcome antibiotic resistance. Trellis’s pan-influenza clinical candidate (TRL404) comprises 3 mAbs that provide broad spectrum: CF-401 x Flu A Group I [includes H5N1], CF-402 x Flu A Group II [includes H7N9], and CF-403 x Flu B [all clades]. Each of these mAbs is as good or better than any published mAb, and the mixture is substantially better than any single mAb. With funding from NIH grants, TRL404 was proven to be highly effective as an acute treatment in mice against both H5N1 and H7N9 (another high pathology avian influenza strain). Published data establish that inhaled delivery provides improved efficacy as compared to delivery by injection. The efficacy window for TRL404 extends to 96 hours post-infection, vs 24-48 hours for currently marketed antivirals. Escape was minimal after passaging the virus 10 times in the presence of TRL404 at a concentration that kills 90% of the virus. Initial commercial quality manufacturing was scaled to support intravenous delivery. Since inhaled delivery reduces the required dose by more than 20-fold, Trellis currently has sufficient bulk drug substance to produce 15,000 doses. The remaining work needed to meet FDA requirements for human testing primarily involves dispensing the bulk drug as drug product and associated bioanalytical assays. With adequate funding, TRL404 should be available as an acute treatment in time to respond to an emerging human H5N1 pandemic.
April 2024
April 30, 2024
Transforming antibody therapy: Trellis Bioscience’s TRL1068 takes aim at antibiotic-resistant infections
(View Article)
In the fight against infectious diseases, such as Prosthetic Joint Infections (PJIs), finding truly effective treatments can be a challenge. Trellis Bioscience, a leading therapeutic antibody company, aims to tackle this issue head-on. Chronic PJIs, where bacteria form protective biofilm layers on artificial joint prostheses, are impossible to treat with antibiotics alone. Trellis Bioscience’s novel antibody, TRL1068, could change the game by removing these layers, offering hope for better PJI treatment and beyond.
March 2024
March 6, 2024
Trellis Bioscience's TRL1068 Receives FDA Fast Track and QIDP Designations, Amplifying Commitment to Combat Infectious Diseases
(View Press Release)
Trellis Bioscience Inc., a pioneering biotechnology company focused on the discovery and development of native human monoclonal antibodies, is thrilled to announce that its lead antibody candidate, TRL1068, has been granted Fast Track and QIDP designations by the U.S. Food and Drug Administration (FDA).
May 2023
Press Release
May 4, 2023
Trellis Bioscience Announces Clinical Proof of Concept in reducing biofilm burden in Prosthetic
Joint Infections with a native human monoclonal antibody TRL1068
(View Press Release)
April 2023
Trellis Pipeline
April 24, 2023
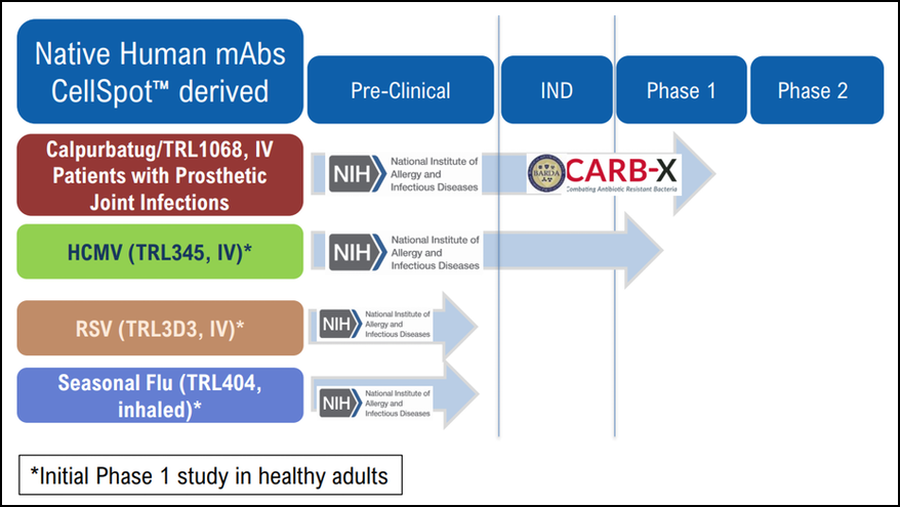 By leveraging its proprietary CellSpot™ platform for discovery of human monoclonal antibodies (mAbs), Trellis has built a pipeline of products
for infectious diseases that pose serious morbidity and/or mortality risks. Our lead product is currently in a Phase 1 clinical trial to treat patients with an infected prosthetic knee or hip joint. In addition, we are developing mAbs against prominent viruses including
human cytomegalovirus (HCMV), respiratory syncytial virus (RSV), and influenza. The mAb segment of the pharmaceutical industry, the fastest growing
class of drugs, reached $185 billion in 2021 with a projected annual growth rate of 11% through 2030. As the cost of mAb manufacturing has declined
over the past two decades, this highly attractive class of drugs has become cost effective for an increasingly wide spectrum of diseases. In particular,
the growing importance of mAbs for infectious diseases was highlighted during the COVID-19 pandemic, with revenues for mAbs against that virus exceeding
$10 billion.
By leveraging its proprietary CellSpot™ platform for discovery of human monoclonal antibodies (mAbs), Trellis has built a pipeline of products
for infectious diseases that pose serious morbidity and/or mortality risks. Our lead product is currently in a Phase 1 clinical trial to treat patients with an infected prosthetic knee or hip joint. In addition, we are developing mAbs against prominent viruses including
human cytomegalovirus (HCMV), respiratory syncytial virus (RSV), and influenza. The mAb segment of the pharmaceutical industry, the fastest growing
class of drugs, reached $185 billion in 2021 with a projected annual growth rate of 11% through 2030. As the cost of mAb manufacturing has declined
over the past two decades, this highly attractive class of drugs has become cost effective for an increasingly wide spectrum of diseases. In particular,
the growing importance of mAbs for infectious diseases was highlighted during the COVID-19 pandemic, with revenues for mAbs against that virus exceeding
$10 billion.
April 6, 2023
Advancing the Clinical Evaluation of Calpurbatug (TRL1068)
Trellis activated the eighth site for its ongoing clinical study of TRL1068-101 (ClinicalTrials.gov Identifier: NCT04763759). The primary objectives of this first-in-human study are to establish the safety and tolerability of calpurbatug in patients with a prosthetic joint infection (PJI), which is difficult to treat due to a biofilm-embedded bacterial infection growing on the artificial knee or hip replacement. Enrolled patients to date have had confirmed infections by a variety of bacterial species, including pseudomonas, staphylococcus, (including coagulase-negative staphylococci), streptococcus, and cutibacterium. The broad-spectrum activity of calpurbatug is thus of high value for this indication, as these bacterial infections are both Gram-positive and Gram-negative. Although designed primarily as a safety clinical trial, the study provides an opportunity to gain initial insight into efficacy in a challenging indication. A single infusion of calpurbatug is given 7 days prior to the scheduled first stage of a two-stage surgery. The first stage procedure is to remove the infected prosthesis, enabling quantification of the biofilm-embedded infection using an established gold standard method. The results will be compared to historical data that used the same methodology. These initial efficacy results will form the basis for a Phase 2 study. Our goal is to spare the PJI patient from the current two-stage surgical procedure and associated quality of life, morbidity and enormous pharmacoeconomic costs. The Phase 1 study is funded in part by a contract with CARB-X (Combating Antibiotic-Resistant Bacteria), a global government and philanthropic partnership to accelerate antibacterial innovation.
April 5, 2023
New Insights into Immune Surveillance Against Cancer
Monoclonal antibodies (mAbs) have proven useful as treatments for both infectious diseases and cancer. The body’s past antibody responses are preserved in the memory B-cell compartment of healthy individuals, providing a mAb source that is particularly attractive although difficult to exploit by prior technology. Trellis’ CellSpot™ platform has overcome the obstacles by providing markedly improved throughput and resolution. This technology enabled Trellis to become the first to discover native human monoclonal antibodies (mAbs) against immune checkpoint modulators (ICMs). In addition, the Company also cloned antibodies binding with high affinity to tumor associated antigens (TAA). While the existence of mAbs against TAAs was previously known, a remarkable new result from Trellis’ work is that mAbs against ICMs are also found in the memory B-cells of healthy individuals, although at a frequency too low for easy detection by prior methods. Several immune checkpoint modulators have been approved for the treatment of cancer, and they have substantially improved cancer therapies. This major advance was recognized by the 2018 Nobel Prize of Medicine. However, it was not previously known that such mAbs are generated naturally. Trellis’s result extends the immune surveillance concept which refers to the immune system’s role in eliminating emerging tumor cells before they cause overt disease. These transiently expressed natural mAbs may be better suited than laboratory generated mAbs for balancing anti-tumor efficacy against safety. Clinical evaluation of the Trellis mAbs will be assessed by our partner Relevant Biosciences, a startup biotech company focused on oncology.
About Relevant Biosciences Inc.
Relevant Bio has a portfolio of novel protein antibody fragment carriers, which were isolated by Trellis’ CellSpot program, for tumor specific targeting ligands with known expression in a variety of cancers,
both solid and liquid tumors and include breast cancer, triple negative breast cancer, myeloma, sarcoma, colorectal, to name a few. In the company’s lead program these carriers are labeled with
either an alpha or beta emitting isotopes depending on the cancer presentation (bulky vs. metastatic disease)
The radiopharmaceutical segment in oncology is rapidly expanding as seen with the > $8 billion acquisition by Novartis of Lutathera and Pluvicto as well as the launch of several built for purpose companies (Aktis Oncology and Curie Therapeutics) which closed Series A rounds at $150 million and $75 million, respectively. Relevant Bio’s management has hands-on experience with radiopharmaceuticals including work on the pivotal study and NDA for Lutathera.
Ryser S, Estellés A, Tenorio E, Kauvar LM, Gishizky ML. High affinity anti-TIM-3 and anti-KIR monoclonal antibodies cloned from healthy human individuals. PLoS ONE 12(7): e0181464 (2017).
April 4, 2023
Structural Insights into How TRL3D3 Neutralizes the RSV G Protein
Trellis’ collaborator at University of California Santa Cruz, Professor Rebecca M. DuBois, will present data on TRL3D3, a native human monoclonal antibody against respiratory syncytial virus (RSV), at the American Society of Virology meeting at the University of Georgia (June 24-28, 2023). X-ray crystallography analysis of the structures of this antibody and a few similar ones provides insights into the conserved conformational epitopes revealed by these high affinity native human antibodies discovered using Trellis’ proprietary CellSpot™ technology. The results highlight the uniqueness of TRL3D3 with preliminary work suggesting that it may one day be feasible to develop a vaccine that can induce antibodies like TRL3D3.
Nuñez Castrejon AM, O’Rourke SM, Kauvar LM, DuBois RM. Structure-Based Design and Antigenic Validation of Respiratory Syncytial Virus G Immunogens. Journal of Virology 96(7):e0220121 (2022)
Bergeron HC, Murray J, Nuñez Castrejon AM, DuBois RM, Tripp RA. Respiratory Syncytial Virus (RSV) G Protein Vaccines With Central Conserved Domain Mutations Induce CX3C-CX3CR1 Blocking Antibodies. Viruses 13(2):352 (2021)
Fedechkin SO, George NL, Wolff JT, Kauvar LM, DuBois RM. Structures of respiratory syncytial virus G antigen bound to broadly neutralizing antibodies. Science Immunology 3(21):eaar3534 (2018)
April 3, 2023
Moving from Prophylaxis to Treatment of RSV Requires a New Approach
Trellis collaborators in the laboratory of Professor Ralph A. Tripp at University of Georgia have recently published two scientific papers that strengthen the preclinical rationale for our novel treatment for respiratory syncytial virus (RSV) infections. Numerous drug candidates targeting the RSV envelope glycoprotein F that have shown efficacy for preventing an RSV infection have failed to show significant efficacy as an acute infection treatment. Native human monoclonal antibody (TRL3D3) offers a new approach. This Trellis antibody targets the other major envelope glycoprotein, the RSV G protein. In preclinical studies, TRL3D3 not only reduces viral load but also neutralizes the airway inflammatory effect caused by the RSV G protein. This is in contrast with the successful prophylactic approaches, including vaccines nearing approval by the FDA, which do not neutralize the proinflammatory activity of the G-protein and thus do not address the most medically significant consequences of an acute RSV viral infection.
Bergeron HC, Tripp RA. RSV Replication, Transmission, and Disease Are Influenced by the RSV G Protein. Viruses 14(11):2396 (2022)
Bergeron HC, Kauvar LM, Tripp RA. Anti-G protein antibodies targeting the RSV G protein CX3C chemokine region improve the interferon response. Therapeutic Advances in Infectious Disease 10:1-16 (2023))
March 2023
March 31, 2023
TRL345: New Insights into Unique Features of this Native Human Antibody
Trellis’ proprietary technology for discovery of native human monoclonal antibodies (CellSpot™) was used to discover TRL345, which binds to the most conserved region of human cytomegalovirus (HCMV). Trellis scientists had previously shown that only a narrow subset of human antibodies is able to bind to this conserved site with high affinity. Academic investigators, working independently of Trellis, have recently verified this result. They concluded that the key somatic mutations responsible for the superior activity of TRL345 represent rare events in the formation of antibodies. This result provides an explanation for the weak neutralizing antibody response to HCMV vaccines to date, which has caused multiple companies to abandon their HCMV vaccine programs, The insightful publication by Jenks et al, an independent academic group, has implicitly elevated TRL345 to the gold standard of HCMV therapy and prophylaxis. The FDA has granted an active IND for TRL345 for a clinical study in healthy adult volunteers.
McCutcheon KM, Gray J, Chen NY, Liu K, Park M, Ellsworth S, Tripp RA, Tompkins SM, Johnson SK, Samet S, Pereira L, Kauvar LM. Multiplexed screening of natural humoral immunity identifies antibodies at fine specificity for complex and dynamic viral targets. MAbs 6(2):460-73 (2014)
Jenks JA, Amin S, Sponholtz MR, Kumar A, Wrapp D, Venkatayogi S, Tu JJ, Karthigeyan K, Valencia SM, Connors M, Harnois MJ, Hora B, Rochat E, McLellan JS, Wiehe K, Permar SR. A single, improbable B cell receptor mutation confers potent neutralization against cytomegalovirus. PLoS Pathogens 19(1):e1011107 (2023)
March 29, 2023
Calpurbatug: Short Summary of Key Preclinical Findings
The therapeutic rationale for TRL1068 (calpurbatug) has been described in scientific publications with academic collaborators at University Hospital
Basel (Switzerland) and Harbor-UCLA Medical Center. Reversal of biofilm-associated resistance to antibiotics has been established in several animal models for both Gram-positive and Gram-negative bacterial infections. The broad-spectrum activity for this native human antibody arises from high affinity binding to a conserved site on a family of
bacterial proteins that hold in place the extracellular DNA that comprises approximately half the mass of the biofilm.
In preclinical toxicology studies, using very high (supratherapeutic) doses of TRL1068 (calpurbatug), the human mAb was found to be safe. This safety profile is attributable to
the features of our proprietary discovery platform Cellspot which enables the cloning of mAbs from human white blood cells of healthy donors. In addition to the fact that the
mAb was safe in at least the human donor, the high-affinity leads to high specificity which typically minimizes potential off-target activity. CellSpot surveys millions of IgG
antibody producing memory B-cells, which means the healthy human donor’s immune system has already evaluated these mAbs for safety. The technology characterizes the binding
specificity of each mAb against 4-6 proteins concurrently, allowing the rare mAbs with broad scope activity to be identified. This computerized microscopy platform yields
approximately 10,000-fold more data relevant to selection of pharmaceutical grade antibodies than prior technologies.
Estellés A, Woischnig AK, Liu K, Stephenson R, Lomongsod E, Nguyen D, Zhang J, Heidecker M, Yang Y, Simon RJ, Tenorio E, Ellsworth S, Leighton A, Ryser S, Gremmelmaier NK, Kauvar LM. A High-Affinity Native Human Antibody Disrupts Biofilm from Staphylococcus aureus Bacteria and Potentiates Antibiotic Efficacy in a Mouse Implant Infection Model. Antimicrobial Agents Chemotherapy 60(4):2292-301 (2016)
Xiong YQ, Estellés A, Li L, Abdelhady W, Gonzales R, Bayer AS, Tenorio E, Leighton A, Ryser S, Kauvar LM. A Human Biofilm-Disrupting Monoclonal Antibody Potentiates Antibiotic Efficacy in Rodent Models of both Staphylococcus aureus and Acinetobacter baumannii Infections. Antimicrobial Agents Chemotherapy 61(10):e00904-17 (2017)
Ryser S, Tenorio E, Estellés A, Kauvar LM. Human antibody repertoire frequently includes antibodies to a bacterial biofilm associated protein. PLoS One 14(7):e0219256 (2019)
INN Names TRL1068 calpurbatug
March 27, 2023
The International Nonproprietary Names (INN) Programme of the World Health Organization announced in March 2023 in their INN List that the generic name of the native human antibody (mAb) TRL1068 is calpurbatug (WHO Drug Information, Vol. 37, No. 1, 2023). The suffix -tug designates unmodified immunoglobulins. This is the first time that a revised nomenclature scheme for monoclonal antibodies specifically recognizes unmodified monoclonal antibodies. The well-known suffix -mab is not available anymore, instead modified immunoglobulins are designated as artificial (-bart), multi-specific (-mig) and antibody fragments (-ment). The root -ba- designates antibacterial immunoglobulins. The root for calpur is California where the antibody was discovered and infused in the first human subject, a patient with prosthetic joint infection (PJI).